Desarrollo de un Método Espectrofotométrico para Analizar el Clorhidrato de Fenilefrina en Forma Pura y Farmacéutica Utilizando Ácido Sulfanílico Diazotizado
##plugins.themes.bootstrap3.article.main##
Resumen
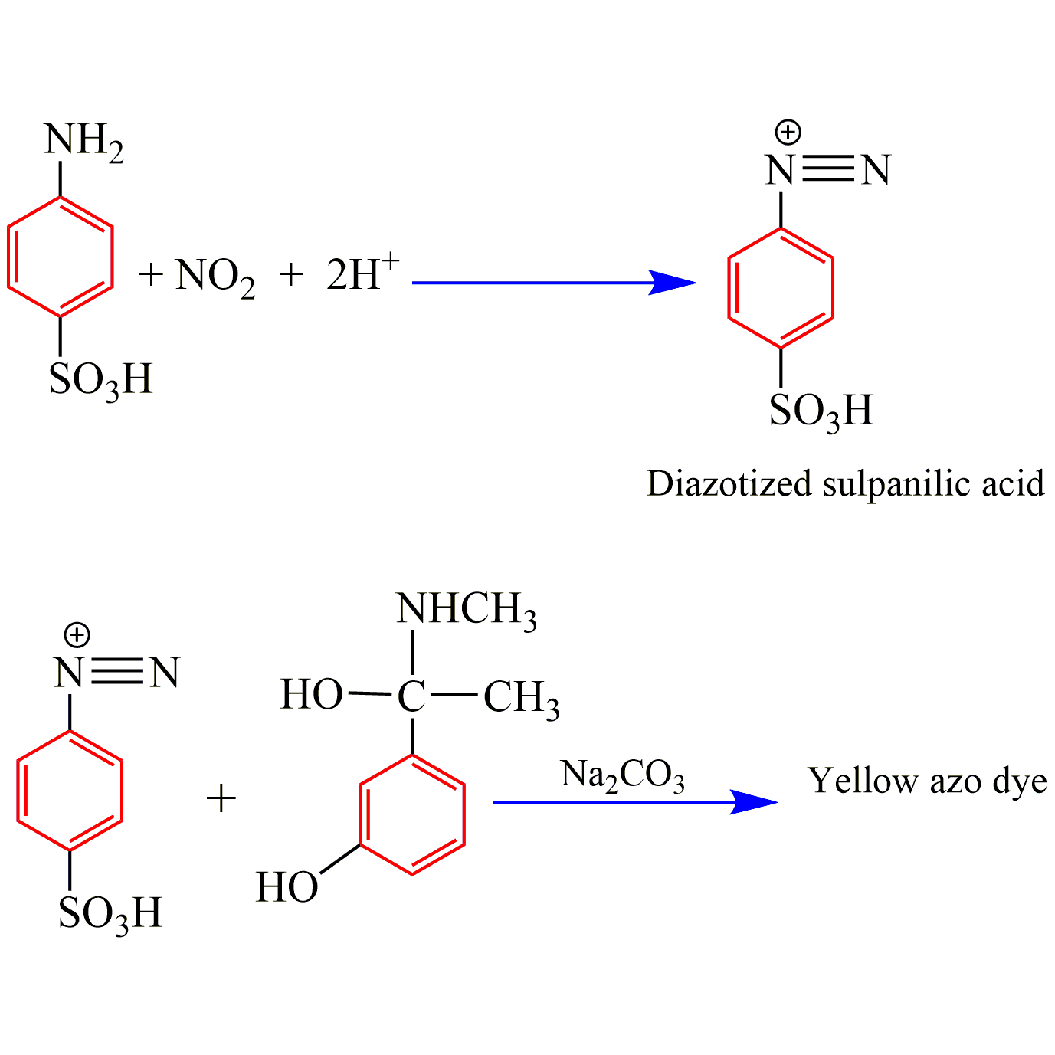
Este trabajo describió un procedimiento espectrofotométrico simple y sensible para estimar el clorhidrato de fenilefrina en estado puro y en sus formas farmacéuticas usando ácido sulfanílico diazotado como agente de acoplamiento en medio alcalino de solución de carbonato de sodio. El colorante azoico amarillo resultante mostró un máximo de absorción a 441 nm. El gráfico de calibración fue lineal y compatible con el mínimo de Beer en el intervalo de concentración de 0.2-20 µg/mL con determinación del coeficiente (r2=0.9996). Los valores de absortividad molar y la sensibilidad del índice de Sandell fueron calculados y fueron de 8.51×103 l/mol.cm y 0 0239 µg/cm2, respectivamente. Los valores del límite de detección (LOD), el límite de cuantificación (LOQ) y el rango del porcentaje de error relativo se estimaron y resultaron ser 0.0050, 0.0166 µg/mL y -0.374 % a -0.147 %, respectivamente. También se calculó la precisión (representada por RSD) y se encontró que estaba en el rango de ±0.037 a ±0.28. El procedimiento sugerido fue útil con éxito para el ensayo de clorhidrato de fenilefrina en inyección, gotas y jarabes. No se observaron interferencias de los aditivos comunes que se encuentran en las preparaciones farmacéuticas. El color amarillo del producto fue muy estable y no mostró cambios significativos en la absorbancia hasta los 60 min.
Descargas
Descargas
Detalles del artículo
Citas
Ahmed, A. Kh.; Anwar, S.M.; Hattab, A.H. (2020). Spectrophotometric Determination of Phenylephrine Hydrochloride in Pharmaceutical Preparations by Oxidative Coupling Reaction, IJDDT, 10(3), 323-327. https://doi.org/10.25258/ijddt.10.3.2
Ahmed, H. H., & Mohammed, S. A. (2023). Spectrophotometric Estimation of Phenylephrine Hydrochloride via Oxidative Coupling Reaction with p- Aminobenzophenone. Biomedicine and Chemical Sciences, 2(2), 83–89. https://doi.org/10.48112/bcs.v2i2.455
Ahmed, I. S.; Amin, A. S. (2007). Spectrophot-ometric micro determination of phenylephrine hydrochloride in pure and in pharmaceutical formulations using haematoxylin, Journal of Molecular Liquids, 130, 84–87. https://doi.org/10.1016/j.molliq.2006.04.011
Al-Abachi, M.Q.; Abed, S. S. (2015). Flow injection-Spectrophotometric Determination of Phenylephrine Hydrochloride and Amoxicillin Trihydrate in Pharmaceutical Preparations, Journal of Al-Nahrain University Science, 16(1), 42-52. https://mail.anjs.edu.iq/index.php/anjs/article/view/711/637
Al-Abachi, M.Q. and Abed, S.S. (2015). Spectrophotometric Determination of Phenylephrine hydrochloride and Salbutamol sulphate drugs in pharmaceutical preparations using diazotized Metoclopramide hydrochloride, Baghdad Science Journal, 12(1), 167-177. https://doi.org/10.21123/bsj.2015.12.1.167-177
Aljeboree, A. M.; Alshirifi, A. B. (2018). Colorimetric Determination of phenylephrine hydrochloride drug Using 4-Aminoantipy-rine: Stability and higher sensitivity, J. Pharm. Sci. & Res., 10(7), 1774-1779. https://www.jpsr.pharmainfo.in
Aljeboree, A.M.; Alshirifi, A.N. (2019). Determin- ation of phenylephrine hydrochloride and amoxicillin in a binary mixture using derivative spectrophotometry methods. IJPQA, 10 (3): 168-177. https://doi.org/10.25258/ijpqa.10.3.10
Al-Shaalan, N.H. (2010). Determination of phenylephrine hydrochloride and chlorpheniramine maleate in binary mixture using chemometric-assisted spectrophoto-metric and high-performance liquid chromatographic-UV methods, Journal of Saudi Chemical Society, 14, 15–21. https://doi.org/10.1016/j.jscs.2009.12.004
Alteemi, H.S. and Kadim, K.H. (2020). Colorimetric Determination of Phenylephrine hydrochloride Drug by Diazotization Reaction, Journal of Physics: Conference Series, 1664(1), 012097. https://doi.org/10.1088/1742-6596/1664/1/012097
Azeez, D.T.; Mohammed, S.A. (2022). Develop-ment of Spectrophotometric Method to Assay Sulfadiazine in Pure and in Pharmaceutical Dosage form through Diazotization and Coupling Reaction. Jou. Raf. Sci., 31(1),23- 34. https://doi.org/10.33899/rjs.2022.172925
Battu, S.; Gandu, V.; Nenavathu, B.P. (2018). Simple spectrophotometric method for estimation of drugs using chloramine-T and indigo caramine dye couple, Asian J Biomed Pharmacies Sci, 10 (69), 19-24.https://doi.org/10.35841/2249-622X.69.6498
British Pharmacopaeia (2022). H.M. Stationary Office, London.
Gary, C. (2004). Analytical Chemistry. John Wiley and Sons, 6th ed., Philadelphia, pp. 90-97. https://chemistrydocs.com/analytical-chemistry-by-g-d-christian/
Delevic, R. (1997). Principles of Quantitative Chemical Analysis. Mc, Graw-Hill, Internatinonal ed., Singapore, pp. 495-502. https://www.amazon.com/Principles-Quantitative-Chemical-Analysis-Robert/dp/0070163626
Hargis, L.G. (1988). Analytical chemistry: principle and techniques. Printic Hall Internationa, London pp. 424-427.
Hasan, S.H.; Othman, N.S.; Surchi, K.M.(2016). Determination of Phenylephrine-HCl Using Conductometric Titration Method. Current Analytical Chemistry, 12(4): 330 – 334. https://doi.org/10.2174/1573412912666151126205443
Hasan, Sh. H.; Othman N.S.; Surchi, K. M.(2020). Using of Diazotized 2,4-Dinitroaniline in Spectrophotometric Estimation of Phenylephrine Hydrochloride, Rafidain Journal of Science, 29(3): 73-64. http://dx.doi.org/10.33899/rjs.2020.166310
Ibraheem, A.K. (2009). Spectrophotometric Assay of phenylephrine hydrochloride Via cuopling with Diazotised p-Nitroaniline, Application to Pharmaceutical Preparation, Tikrit Journal of Pharmaceutical Sciences, 5(2), 182-191. https://www.iasj.ne
International Conference on Harmonization (2005). ICH Harmonized Tripartite Guide-line: Validation of Analytical Procedures Text and Methodology, Q2 (R1), Current Step 4 Version, pp. 11-13.
Mzban, Q.; Bahja, S.; Hassan, M.J.M. (2020). Dispersive liquid-liquid microextraction and spectrophotometric Determination of cefazone and phenylephrine hydrochloride in their pure forms and pharmaceutical preparations, Plant Archives, 20(2), 6771-6777. http://www.plantarchives.org/20-2/6771-6777%20(6664).pdf
Nagarjuna, P.J; Kumar, B.T.; Nalluri, B.N. (2020) Simultaneous Analysis of Phenylephrine HCl and Ketorolac Tromethamine in Bulk and Injectable Formulations by RP-HPLC-PDA Method, Journal of Drug Delivery and Therapeutics 10(4s), 39-45. https://doi.org/10.22270/jddt.v10i4-s.4204
Nejres, A. M., & Najem, M. A. (2022). Use of Mesalazine for the Determination of Dopamine and Its Pharmaceutical Preparations by Spectrophotometric Method. Israa Univ. J. Appl. Sci, 6(1), 228-245. https://doi.org/10.52865/AVWR7365
Othman, N.S.; Abdul Fatah, N. Th. (2009). Spectrophotometric Determi-nation of Phenylephrine Hydrochloride by Coupling with Diazotized 2-Aminobenzothiazole, Jou. Raf. Sci., 20(4), 69-81. http://dx.doi.org/10.33899/rjs.2009.39971
Radhia, N. D.; Alshamusi, Q.K.M.; Sahib, I.J.; Jaism, L.S.; Aljeboree, A.M. (2021). Oxidative coupling of phenylephrine hydrochloride using N,N-dimethyl-p-phenylenediamine:Stability and higher sensitivity, 3rd International Scientific Conference of Alkafeel University, 030026-1. https://doi.org/10.1063/5.0066984
Ragab, M.A.A.; Abdel-Hay, M.H.; Ahmed, H.M.; Mohyeldin, S.M. (2019). Determination of Ibuprofen and Phenylephrine in Tablets by High-Performance Thin Layer Chromatography and in Plasma by High-Performance Liquid Chromatography with Diode Array Detection, Journal of Chromatographic Science, 57(7), 592–599. https://doi.org/10.1093/chromsci/bmz031
Suratiya S.; Pancholi, S.S. (2014). Development and validation of dual-wavelength method for Simultaneous Estimation of Ebastine and Phenylephrine hydrochloride in Bulk and Pharmaceutical Dosage Form. International Journal of Pharmamedix India, 2(3):781–791.
The United states Pharmacopoeia (2008). USP 32 NF 27, by Roydan, USA, p.3284. https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/USP32-NF27SecondSupplementRevisionCommentary.pdf
Wasan A. Al-Uzri (2019). Determination of phenylephrene hydrochloride in pharmaceutical preparations using spectrophotometric method, Asian J Pharm Clin Res, 12(5), 339-343. https://doi.org/10.22159/ajpcr.2019.v12i5.32339
Yagmur, S.; Ture, M.; Saglikoglu, G.; Sadikoglu, M.; Yilmaz, S.(2018). The Quantitative Detection of Phenylephrine in Pharmaceutical Preparations and Spiked Human Urine by Voltammetry, Russian Journal of Electrochemistry, 54, 741–746. https://doi.org/10.1134/S1023193518100063
Zakaria, S. A. (2021). Simple spectrophotometric method for Determination of phenylephrine hydrochloride in pure and pharmaceutical forms, Iraqi National Journal of Chemistry, 21(1), 19-29.